This blog was written by Bill Marshall, CEO of miRagen Therapeutics, as part of the “From the Trenches” feature of LifeSciVC.
The opportunity to use nucleic acids as therapeutic agents is extremely exciting. Creating drugs that affect protein targets that are considered “undruggable” by small molecule or protein therapeutics, RNA transcript alteration and up regulation or replacement, systems biology manipulation, even genome correction / alteration are amongst the possibilities.
Nucleic acids as a drug modality encompass a vast array of approaches with a set of key considerations based on their size and mechanism of action. I want to concentrate on synthetic nucleic acid therapeutics, with a primary focus on oligonucleotide therapeutics. Examples include aptamers, antisense, small interfering RNA (siRNA), exon skipping, RNA editing, microRNA therapeutic inhibitors (antimiR) and mimics (promiR), long non-coding RNA modulators and mRNA.
By just the length of that list, it’s clear that it’s an exciting time for the field. In addition, the field has witnessed FDA approval of the first systemically administered therapeutic, significant clinical developments in many areas, continued enabling technology development and an ever expanding “RNA world” allowing for new targets and applications. With continued investment and a focus on technological improvements allowing for more effective and safe drug candidates, the future of the modality looks very promising.
Key considerations in nucleic acid drug development
Nucleic acid drug development has requirements that are the same as any other modality and some that are unique. The usual factors are obvious: drug candidates need significant potency, stability in biological systems, favorable pharmacokinetics (PK), distribution and pharmacodynamics (PD), acceptable specificity, and safety. But nucleic acid drugs fall somewhere between small molecules and biologics and create a new set of considerations. The target tissue, size of the drug molecule, and it’s mechanism of action drive requirements for chemical modification and delivery strategies.
Selection of indications where small molecule or protein therapeutics have had limited success is often a primary consideration. Administration to tissues and cell types that are amenable to the PK/PD of the nucleic acid moiety or that allow for localized administration is another factor. As a result, many applications have focused on targets in the liver, kidney, eye and skin. As we continue to optimize the potency and safety of the drugs, and understand the amenability of additional cell types for productive uptake, the landscape will expand.
For intracellular-directed nucleic acid drugs, productive uptake into the appropriate tissue, cell type, and compartment inside the cell is vital. Nucleic acid molecules are charged and larger than traditional small molecules, so productive uptake into target organs and cells is often challenging and a huge factor in the success of a drug candidate. Given this, careful consideration needs to be given to the size of the nucleic acid drug and the range of chemical modifications that are allowable and still support the mechanism of drug action.
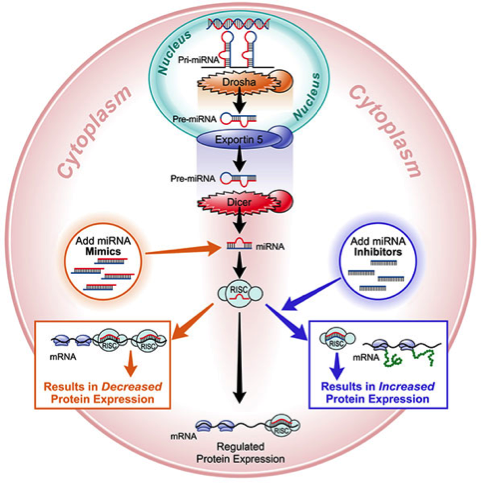
Small interfering RNAs are double-stranded RNA analogs with a molecular weight of about 13,000 – 16,000 (two 22-27 nucleotide strands) with 40+ negative charges. Their mechanism of action requires that they enter a protein complex known as the RNA Induced Silencing Complex within the cell, which then causes the degradation of a messenger RNA (mRNA), resulting in gene silencing. The requirement for these drugs to use cellular machinery for their mechanism of action limits the amount of chemical modification that can be deployed since specific proteins in the cell need to recognize the molecule. The resultant drug molecules thus have core requirements that cannot be altered to modulate other medicinal chemistry properties. For these molecules, delivery strategies like conjugates or formulations are required to help with PK properties and/or facilitate effective cellular uptake. microRNA mimics are also double-stranded and have similar requirements.
Antisense oligonucleotides are single-stranded nucleic acid analogs that also silence genes by targeting individual mRNAs. They are typically 14 to 20 nucleotides with molecular weight of 4,000 – 6,000 and 13-19 negative charges. They take advantage of another endogenous cellular mechanism, this time using RNase H that is more permissive to chemical modification. Thus the core active drug molecule can be altered with hydrophobic and affinity-enhancing modifications that improve medicinal chemistry properties. While there is more freedom in modification, the cellular RNase H activity requirements impose restrictions on where sugar modifications can be introduced in the molecules, limiting them to their outer flanks (constructs known as gapmers). In this case, formulation or conjugation is not required for PK or cellular uptake; although conjugate approaches may improve targeting and uptake, which can be dose sparing.
microRNA inhibitors or antimiRs are single-stranded nucleic acid analogs that bind specifically to a particular microRNA. Because microRNAs regulate multiple mRNAs, antimiRs affect the expression of entire gene networks, which is what gets us excited about their potential at MiRagen. The nature of the interaction between a microRNA and its target mRNAs permit shorter length nucleic acid analogs, and therapeutic antimiR candidates are typically 8 to 15 nucleotides with a molecular weight from about 2,400 – 5,000 with 7 to 14 negative charges. In addition, antimiRs do not require cellular enzymatic activity, so steric interference alone is sufficient as their mechanism of action. This broadens the amount and location of sugar modification, which can be introduced throughout the molecule (constructs known as mixmers). This provides additional opportunities for optimization of the core drug molecule for medicinal chemistry properties such as PK and cellular uptake. Again, conjugates or formulations that can enhance targeting and uptake in tissues would be beneficial, but are not required.
The oligonucleotide gapmers and mixmers contain phosphorothioate modifications to their internucleotide bonds that provide resistance to nucleases and increase the molecule’s hydrophobicity. The change to their charge character via substitution of a sulfur for an oxygen leads to non-specific interactions with serum proteins and tissues, resulting in improved distribution to a range of tissues after systemic administration.
On the other end of the spectrum is synthetic messenger RNA. This approach must deliver the mRNA coding sequence for a gene of interest intracellularly, resulting in expression of the protein for which the gene encodes. The average length of a eukaryotic mRNA is between 1,500 and 2,000 nucleotides with resultant molecular weight of 450,000 – 600,000. Some specific modifications to the mRNA sequence and structure are allowable and can improve the stability and activity of the mRNA, but the mechanism of action requires the cellular translational apparatus to recognize the drug molecule. Because of the macromolecular nature of these molecules and their intracellular mode of action, formulation will be essential for effective delivery.
Maturation of the field, building on decades of experience
I hope the “geeky” section above didn’t scare too many people away! My point was to drive home the fact that there are unique requirements and challenges for each type of synthetic nucleic acid therapeutic approach. Many observers to the field lump them together and make broad-based conclusions on the entire area based on results reported for one approach. However, if you only consider the molecular weight range for the various molecules (2,400 – 600,000), one realizes that it covers a broad spectrum of sizes spanning the known drug universe from small molecules to antibodies and beyond.
For synthetic oligonucleotide therapeutics, recognition of the unique attributes of each class and subsequent application of specific solutions for each has contributed to significant advances in the field. Applying tailored solutions based on the characteristics of the molecules rather than attempting to find a universal solution for targeting and cell uptake (which likely doesn’t exist) is the preferred path. My view is that if we can identify safe, effective molecules that are able to enter their target tissues and cells based on their molecular properties, we can continue to progressively de-risk each approach in humans as we continue to optimize delivery solutions.
siRNA delivery technology has been an area of significant investment. In many cases it has leveraged prior work, which was focused on non-viral approaches to gene delivery, so this is a body of work focused over many decades. Nanoparticle delivery of double-stranded RNA has led to preliminary clinical success, particularly in delivery to the liver. Multiple clinical trials are underway and mechanistic proof of concept for gene silencing delivered by the approach has been obtained in humans. Work in this area continues and with ligand targeting approaches may allow for an expansion into additional tissues.
The development of conjugate delivery approaches is very exciting and significant clinical success has been achieved with GalNAc attachment to siRNA for liver delivery. It is interesting to note that while the current generation of GalNAc conjugates are a relatively recent development, the idea of using ligands for the asialoglycoprotein receptor was contemplated since the late 1980’s, thus again leveraging a history of investigation in the field towards a specific solution. The continued development of conjugate delivery approaches to other tissues and cell types can have implications for more effective drugs for all the oligonucleotide therapeutics.
Single-stranded oligonucleotide therapeutics
Antisense oligonucleotide therapeutics are the most clinically explored of the nucleic acid therapeutic class, and have a long history of discovery and development. The concept of using a short single-stranded oligonucleotide to bind to an mRNA and inhibit its expression was first proposed in the mid 1970’s. Initial work focused on short unmodified DNA oligonucleotides, but the instability of these molecules in biological systems led to a furious effort to identify stable analogs. To make a long story exceedingly short, years of effort led to the primacy of the phosphorothioate internucleotide bond and certain 2’ sugar modifications as the primary oligo therapeutic pharmacophore.
The evolution of antisense technology moved from DNA (1st generation) to hybrid 2’-alkyl modified RNA / DNA hybrid molecules (2nd generation) and onto 2’-constrained bicyclic RNA / DNA hybrids (3rd generation). Constrained nucleoside derivatives, such as Locked Nucleic Acid (LNA) and Constrained Ethyl (CET), allow for a significant increase in absolute potency of target engagement, stabilize oligonucleotides in biological systems, and also positively affect cellular uptake. The mechanistic basis for this effect on productive uptake into cells isn’t completely understood, but empirical analysis has been employed to identify more effective drug candidates.
The promise of antisense as a broad based platform to target any gene and one that could rapidly identify drug candidates led to the notion that any oligonucleotide therapeutic is identical in its pharmacological potential. This was based on being able to identify an accessible site on the targeted mRNA. However, it is clear that a host of factors independent of target engagement play a role in the broader pharmacology of oligonucleotides. We now know that not every 2’-LNA modified phosphorothioate oligonucleotide is created equal.
Because oligo drugs are short polymers of nucleosides, they are typically noted as text strings (5’-ATGCAGTCGATCAC-3’) which imparts a further misplaced sense of uniformity. In reality, these short oligomers have unique structures that impact their biophysical properties. What we canonically view as minor changes to an oligonucleotide can actually lead to profound changes in the pharmacological properties of the molecules. Multiple investigators have reported substantial differences in the safety and productive uptake of this class of molecules based on modest alterations to sequence or modification (one example here).
A recent publication reported on quantum mechanical modeling of short, model oligonucleotides. The quantum mechanical calculations allow for the generation of structures and electrostatic surface potentials for the molecules, which are primary determinants of interaction between molecules. The modeling provides evidence that very conservative changes in the oligonucleotide can lead to profound changes in structure and electrostatic surface potentials. These significant changes in structure and surface potential provide a viable linkage for explaining the substantial differences observed in the behavior of these molecules, especially as relates to complex pharmacological properties such as toxicity, protein binding and tissue and cell uptake.
Moving forward with the latest generation of high affinity single-stranded oligonucleotide drug candidates, the process has evolved into themes common to small molecule candidate selection. Screening campaigns designed to identify a core pharmacophore, optimization of cell and tissue uptake, final candidate selection based on a balance of potency and safety.
In the case of gapmers the length and chemical modification pattern is more fixed and screening involves making sequences that span the length of the targeted mRNA. Potent lead molecules that show uptake and target engagement in vitro and in vivo are then screened in exploratory toxicology to identify the best molecules with good distribution, potency and safety to move forward into clinical trials.
In the case of mixmers, which act through steric interference alone, the sequence space available for exploration is often quite limited. This is most certainly the case for antimiRs where the target microRNA is roughly 22 nucleotides in length. In this case the screening exercise involves varying oligonucleotide lengths, the amount of LNA and DNA in the molecule, and the patterning of LNA in the candidate molecules. Leads are selected based on productive uptake and target engagement and final candidate selection based on exploratory toxicology.
While first and second generation molecules have been primarily focused on applications locally or in the liver, third generation oligonucleotides have a combination of potency, biodistribution, cellular uptake and safety that allow for expansion into other tissues and cell types. The screening paradigms mentioned above have allowed for the identification of molecules with significantly enhanced potency and therapeutic indices that allow for the expansion.
RNA therapeutics are emerging as another major drug modality. Much like the development of the antibody therapeutic modality, the road to success has been winding and marked by several bumps. For antibodies, the technological advancements had to do with better screening for high potency molecules and the development of humanization methodology. The most important analogy is that the promise of the technology forced initial suboptimal molecules to be used for clinical evaluation. A subsequent series of technological advancements occurred that improved potency and safety allowing for expansion of the promise of the modality. Nucleic acid drugs will likely follow a very similar path.
There is certainly an impressive pipeline of drugs based on using complementary nucleic acids as a pharmaceutical agent, suggesting that there is the potential for multiple drug approvals as programs mature. Because of the pharmaceutical product development lifecycle, many molecules in later stage development have not benefitted from the latest technological improvements. This suggests that the full potential of the modality will become more apparent as the newer generation molecules mature. With the advent of non-coding RNA manipulation, the landscape of addressable drug discovery opportunities also expands. My hope is that the technology will continue to prove itself as clinical trials read out and that the field can deliver on the promise of bringing important medicines to patients in need.