This blog was written by Kevin Pojasek, co-founder, President, and acting CEO of Quartet Medicine, as part of the “From the Trenches” feature of LifeSciVC.
Addressing unmet need in chronic pain
In the mid-1990’s, the American Pain Society and the Veterans Health Administration declared pain as “the fifth vital sign” to elevate physician awareness and to highlight the need for better pain reducing treatments.  Despite this increased awareness, there have been no new drugs based on novel mechanisms of action added to the physician’s arsenal of therapies for chronic pain in the intervening 18+ years. Nonetheless, the worldwide market for chronic pain treatments exceeds $25B underscoring the dramatic prevalence of chronic pain.
Despite this increased awareness, there have been no new drugs based on novel mechanisms of action added to the physician’s arsenal of therapies for chronic pain in the intervening 18+ years. Nonetheless, the worldwide market for chronic pain treatments exceeds $25B underscoring the dramatic prevalence of chronic pain.
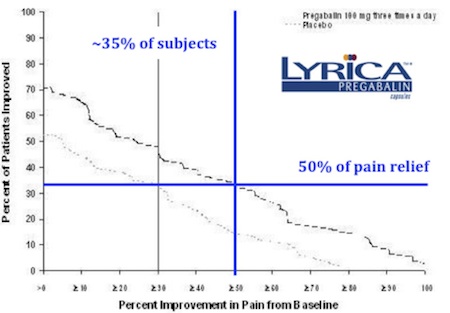
Remarkably, though, best-in-class chronic pain therapies, such as Lyrica (pregabalin), only provide ~50% pain relief to about a third of patients taking it for neuropathic pain conditions, such as painful diabetic peripheral neuropathy (DPN) and post-herpetic neuralgia (PHN).
This astoundingly low level of efficacy is combined with significant side effects – somnolence, weight gain, and dizziness impact up to ~30% of patients on Lyrica. The lack of effective pain drugs based on modern, breakthrough science has also driven the rise in chronic opioid use, a drug class that has been around since ancient Greece. The opioid addiction and overdose epidemic that is sweeping parts of the US today is being fueled in part by this dearth of new treatment options.
At the same time, development of novel analgesics is notoriously challenging – animal models aren’t predictive, subjective clinical endpoints result in high placebo response rates, and the list goes on. Clinical failures have driven many frustrated large and small pharma companies from the space. In recent years, however, those companies still engaged in the battle to identify new options , such as Neusentis (Pfizer), Genentech, Teva, Convergence, and Spinifex have been doing an admirable job of advancing novel pain treatments based on new mechanisms into early clinical trials.
A more recent theme in analgesia research has been to focus on targets in the peripheral nervous system. The premise is that by disrupting the aberrant nerve firing, you can stop the spontaneous pain experienced by many patients with chronic neuropathic pain. Peripheral targets, such as NaV1.7 and the NGF pathway, could enable synergy with existing pain medications that act at the level of the central nervous system (CNS), and also deliver a more desirable safety profile. In fact, NaV1.7 antagonists have started to show clinical promise in a few indications, such as trigeminal neuralgia and lumbosacral radiculopathy. At the same time, the jury remains out on whether the state-dependent binding of most the small molecules will have the horsepower to treat “slower firing” neuropathies, such as PHN and painful DPN. Antibodies to NGF have also shown clinical promise for treating chronic pain in diseases like osteoarthritis. However, Pfizer’s tanezumab program was placed on clinical hold due a safety signal related to acceleration of cartilage destruction in some patients taking the drug. Spinifex, a fellow venture capital-backed biotech, recently demonstrated impressive clinical efficacy in PHN with their angiotensin II Type 2 receptor antagonist program.
Enter tetrahydrobiopterin (BH4)
We set out to build on the theme of peripheral nerve targets by looking at new breakthrough biology at the nexus of the peripheral nerve system and the immune system.
De novo production of tetrahydrobiopterin (BH4) is significantly increased in injured or hyperactive peripheral nociceptors, as well as in a variety of immune cells, including T cells, macrophages and dendritic cells. As a small molecule cofactor for amino acid hydroxylases and nitric oxide synthases, increased levels of BH4 are thought to have a pluripotent effect on a variety of downstream pain mediators. Safely restoring BH4 levels back to baseline by targeting one of the key BH4’s synthetic enzymes, either sepiapterin reductase or GTP cyclohydrolase I (GCH1), has been shown to reduce inflammation and painful hypersensitivity in preclinical models.
There are several features that make the stress- and injury induced de novo BH4 pathway an especially appealing target for treating pain:
- Relevance to human disease – Data from Clifford Woolf’s lab first demonstrated that a specific human GCH1 haplotype correlates with less chronic pain after injury and an inability to elevate BH4 levels during nerve stress (despite these haplotype carriers having normal baseline BH4 levels). Other labs have now replicated these original findings. And while the link to BH4 is not ubiquitous to all painful conditions, it does appear to play a critical role in the chronification – the progression of acute to chronic pain — after injury and disease.
- Separately, Kai Johnsson discovered sulfasalazine and other sulfa drugs are nM inhibitors of sepiapterin reductase and BH4 synthesis. Sulfasalazine’s status as a decades-old, disease-modifying, antirheumatic drug for treating rheumatoid arthritis and ulcerative colitis further ties in the clinical relevance of BH4 in human disease. At the same time, sulfasalazine has significant liabilities – daily dose of 4 grams, highly variable absorption and exposure, and drug class-related side effects to name a few – that limit it’s development as a chronic pain treatment.
- Confidence in safety – BH4 is a widely-produced and requisite enzyme cofactor; complete depletion of BH4 would impact both CNS and cardiovascular (CV) function. Importantly, carriers of the GCH1 haplotype do not appear to be at undue risk for CNS or CV-related disease. In addition, patients taking high-dose sulfasalazine experience only modest CNS-related side effects with no increase in blood pressure or other impairments of CV function.
- Biomarker for target engagement – Having a peripheral measure of target engagement is unusual for a chronic pain program. The ability to nucleate an R&D strategy around correlating drug exposure with a reduction in BH4 in the peripheral nerve tissue and immune system before advancing compounds into notoriously unpredictable preclinical models is an attractive advantage for this approach. The BH4 biomarker will also be a valuable tool for assessing objective target engagement as the program advances into clinical trials with inherently variable patient-reported outcome endpoints.
- Tractable drug discovery – Excellent screening work done by the Johnsson Lab yielded a diverse and potent array of small molecule inhibitors of BH4 synthesis entirely distinct from sulfa-based drugs, such as sulfasalazine. Access to this information, along with robust in vitro drug discovery tools, has likely accelerated the program by ~18 months.
Assembling and launching Quartet
Working closely with Atlas partners, Bruce Booth and Dave Grayzel, as well as our academic founders, Clifford Woolf and Kai Johnsson, we assembled the key intellectual property to support the founding of Quartet Medicine late last year. Atlas invested ~$900k in seed capital to validate the supporting preclinical biology, initiate drug discovery efforts, assemble a team, and explore market interest in our program. We elected to partner with Evotec early on to advance the program through a creative, risk-sharing partnership structure.
The data generated during the seed phase confirmed our enthusiasm for the pathway. We closed an oversubscribed $17M Series A adding Henry Skinner and Christine Brennan from Novartis Venture Funds, Elaine Jones from Pfizer Venture Investments, and Jay Knowles from Partners Innovation Fund to the Atlas team in the Quartet board room. We’ve also attracted top-notch drug discovery executives to the Quartet management team, adding Annika Malmberg as our head of biology and Mark Tebbe as our head of chemistry. Alasdair Naylor and Eddine Saiah have also been integral contributors to advancing our ongoing drug discovery efforts.
Assembling Quartet has been one of my main projects while working as an Atlas entrepreneur-in-residence. I’m proud of the progress we’ve made to date and look forward to what lies ahead as we graduate from the Atlas “seed pool”.