This blog was written by Bill Marshall, co-founder and CEO of miRagen Therapeutics, as part of the “From the Trenches” feature of LifeSciVC.
microRNAs are an exciting new class of gene regulatory molecules that act on systems biology, and their modulation represents an attractive opportunity to treat complex diseases that have been difficult to address with more traditional approaches. By using a combination of genetic and pharmacological studies, we’ve been able to validate the role of specific microRNAs that appear to be very attractive drug targets.
It has been very exciting to work on the cutting edge of biology and approach a new modality for therapeutic intervention that is only beginning to be explored. At the same time, the usual uncertainties about the target and drug are amplified in a new paradigm. As we approach the clinic we’ve tried to maintain a cutting edge approach to science in our translational medicine program.
Beachhead Clinical Development Approach
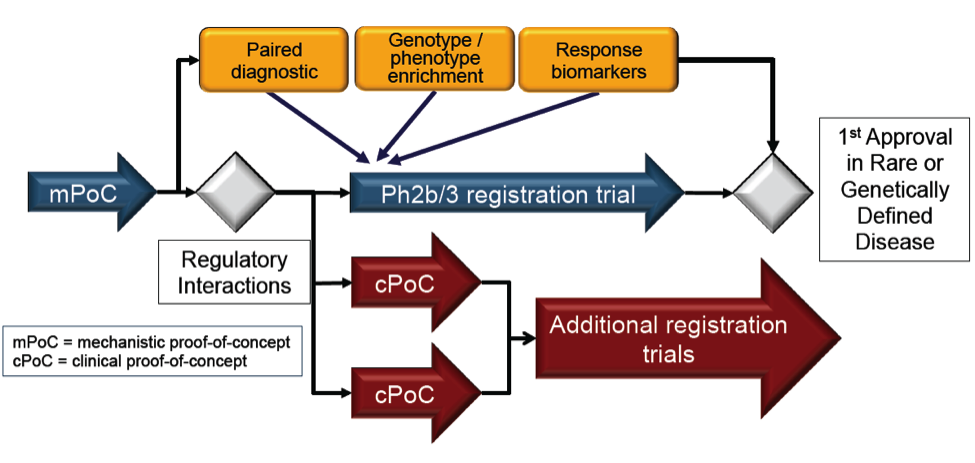
Our “beachhead” approach to translational medicine is focused on rapidly testing the molecular hypothesis for drug action in humans by taking advantage of exploratory IND (eIND) studies that were first outlined in the US FDA Critical Path Report in 2004. These studies are conducted early in phase 1, involve limited systemic exposure, and have no therapeutic intent.
We term these beachhead because they are designed to test the mechanistic relevance in humans in a setting that provides the opportunity to develop a biomarker toolkit for a core mechanism of action that we believe has broader disease relevance. We therefore attempt to identify a clinical condition that is amenable to local administration and tissue sampling, but whose molecular basis has links to other disease indications.
The goal is to progress rapidly to first in human studies once we have established pharmacokinetics / pharmacodynamics and shown that the drug candidate is safe in preclinical studies. The beachhead indication is typically a genetically defined or rare disease where dysregulation of a microRNA has been implicated.
The mechanistic proof of concept (mPOC) studies provide a host of relevant information that helps to progressively derisk the target / drug pair in humans. Our aim is to demonstrate that the expression levels of the microRNA itself could potentially serve as a companion diagnostic which allows for patient enrichment in later clinical trials and in additional indications. At the same time we can validate a set of pharmacodynamic markers of target engagement and surrogate markers of drug action.
By measuring the pharmacodynamics of target engagement we are able to show that the drug candidate effectively enters the appropriate cell and binds to its intended target. This is particularly important for oligonucleotide based drugs. We can also measure the effects on a series of downstream genes that create a plausible link between target engagement and a mechanism that could be important to clinical benefit.
Importantly, the local administration allows us to achieve a variety of concentrations of drug at the site of action and facilitates the development of dose / response relationships. Understanding the dose necessary to show target engagement with concomitant surrogate marker alterations provides the foundation for establishing the systemic dose that will be necessary to potentially achieve a therapeutic effect.
With a biomarker tool kit and dose guidelines in hand from the eIND studies, the traditional multiple ascending dose phase I study is conducted with primary endpoints of safety and pharmacokinetics. Exploratory endpoints can then give us verification of pharmacodynamics with systemic administration as well as surrogate biomarker readout and morphological alterations.
This translational strategy allows one to answer many fundamental questions about the drug target pair and provide confidence that the molecular basis of drug action is relevant in man. After all, this is obviously the most important and relevant species for this endeavor!
I believe that there is limited value in spending much time using animal disease models of efficacy as tools and certainly not to gate decisions to move forward into clinical evaluation. Unless the model system truly mirrors the drivers of human disease and has been shown to have significant predictive value its use only “makes us feel better.” An unbiased view of the value of animal models suggests that they have little positive or negative predictive value. We therefore use animal models primarily for insights on pharmacokinetics and pharmacodynamics and in safety and tolerability studies.
Our approach to early translational proof of concept is something that we have adapted from the pioneering work of many others in the pharmaceutical world, especially Novartis. My colleague Dave Rodman was part of the original translational medicine group assembled by Novartis to rethink the way we move medicines forward in man and is responsible for many of the guiding principles outlined here.
Improving the probability of success through early proof of mechanism studies
There have been many analyses that have pointed to potential solutions to the Pharma R&D productivity crisis, especially as it relates to small molecules (here, here, here). One common theme is better target selection / validation and performance of early proof of concept studies in man.
A publication by investigators at Lilly provided an interesting model of the drug discovery process and pointed out these two features as being vital to improving attrition of drug candidates in phase II and III. Their analysis suggests that a combination of better target selection and phase I proof of concept studies could improve the rate of success for moving from phase II to phase III from 34% to 50%. They estimated that this alteration alone could reduce the cost of a new molecular entity by approximately 30%.
For several microRNAs, there is a wealth of validation provided through genetic studies and associations to human genetics, which provides a solid basis for target validation. The application of phase I proof of mechanism studies in man to non-traditional drug modalities such as microRNAs should help to improve the success rate as we attempt to move these approaches from the realms of science fiction to life changing medicines.