With the big cancer confab ASCO 2016 fading into memory, R&D teams everywhere are getting back to work on their cancer programs – and across the industry many of them will be doing the same work, on the same targets, supporting the exploding supernova known as immuno-oncology.
The rapid expansion of this space in the past five years has been exceptional to witness: catalyzed by striking clinical data, reflecting real changes in the survival curves of an ever-broader set of cancers, a huge number of I/O programs have advanced across the industry, fueled by prodigious amounts of capital. This is in many ways great news for patients and the industry.
But it does raise long term strategic and capital allocation questions for cancer R&D. This blog has raised this topic a few times in the past five years. In a 2012 post, The March of the Lemmings, the a huge amount of work on a small set of kinase targets was highlighted – including 70 programs on VEGF/VEGF (here) – after PhRMA shared a snapshot of the industry pipeline 900+ oncology programs. In 2015, a quick review of ASCO’s abstracts revealed the dominance of either ErB family members (e.g., EGFR, HER2, etc) or immuno-oncology checkpoint targets (here). BioCentury did a similar chart review of the abstracts in 2016 last month (here), with the same expected findings: the abstracts were dominated by I/O related targets. Now even the FDA’s oncology czar, Richard Pazdur, has raised questions about the explosion of “me-too” I/O drugs in the cancer field (here, here), in a dialogue with Reuters at ASCO this year.
Rather than write in general about this topic, I wanted to explore two things: frame the actual scale of the issue today, and then touch on some of the strategic ramifications for pharma, biotech, and the venture ecosystem.
Scale of Activity
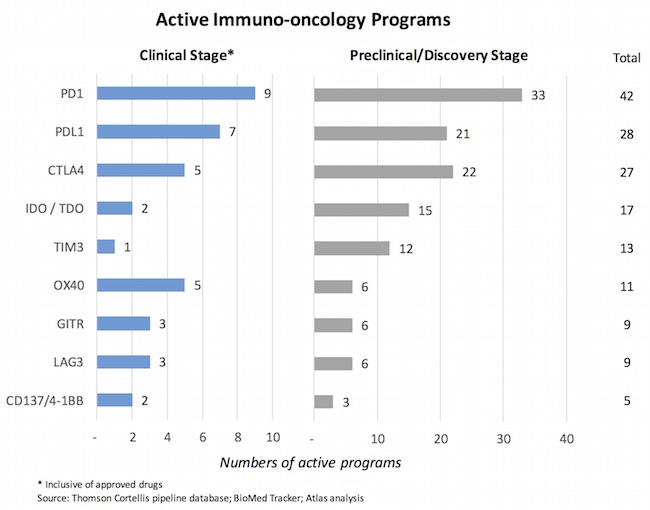
So how many programs against the key checkpoint targets exist in R&D today? It’s been only five years since the first checkpoint inhibitor, anti-CTLA4 Yervoy (ipilumumab), was approved for melanoma in 2011. My colleague Steve Robinette cranked up the analysis below by looking at a few datasets. I’m sure this is missing some programs, but is certainly directionally correct. By our count, there are at least 16 discrete PD1/PDL1 programs in the clinic, and 70 if you include those disclosed but in pre-clinical stages. Several of these are in the hugely capital-intensive Phase II-III clinical trial window before approval. In addition, there are more than two dozen CTLA-4 programs are being pursued, and a scores of projects against other “emerging” checkpoints like TIM3, LAG3, OX40, etc…
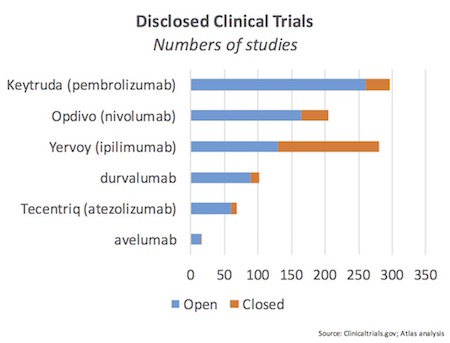
Just taking a look at the four approved checkpoint inhibitors and two late-stage PDx programs, there are nearly 1000 studies registered with ClinicalTrials.gov, the vast majority of which are open currently. Keytruda alone or in combination has nearly 300 studies. And these include only those clinical studies that have been disclosed/reported; I’m confident there are many more investigator-initiated trials done at academic medical centers that have yet to be disclosed.
If you want some additional visuals, check out this Citeline interactive website, or this “bullseye” chart from twitter (here) – both reinforce the hyper-active scale of the I/O space today.
Furthermore, these charts don’t capture the “other” I/O settings – like chimeric antigen receptor T-cell therapy (CART), where a dozen or more programs are in development targeting the same B-cell marker, CD19, in various forms of leukemia (where the majority of the compelling CART data exists today) and lymphoma.
Based on the data above, and affirming Pazdur’s sentiments, it’s strikingly clear that a huge amount of (potentially redundant) investment and effort is focused on chasing the same set of cancer immunotherapy targets.
Strategic Issues
This flood of I/O projects presents a large number of important strategic implications; it’s very hard to do justice to them all with one blog post, but here are four top of mind thoughts:
First, this wave of programs face a massive product differentiation challenge. To date, Opdivo has beaten Keytruda in the marketplace, in large part driven by the latter’s requirement for a PDL1 expression test before prescribing (here). But Keytruda is building a huge clinical program. Both are clearly “here to stay” as foundational therapies. With these two entrenched PD1s, and Roche’s recent approval of Tecentriq (PDL1), how will the others – like durvalumab, avelumab, and the other dozen PDx’s in the clinic – differentiate from each other? For now, most clinicians think Keytruda and Opdivo deliver largely similar efficacy, with nuances around dosing and who has what clinical data in their label, etc… But will all the PDx’s and CTLA4s (and various combos and doses/schedules therein) behave the same in most settings? If they don’t, how will this be proven? These PDx anitbodies likely have a range of epitopes, affinities, Fc functions, glycosylations, etc – all of which could affect activity, but differential activity isn’t likely predictable from preclinical work. And in the real world setting many of the nuanced differences may disappear in the face of disease complexity. My guess is that it will take many clinical studies and lots of patients to prove one is better than the other – which will require is a gargantuan effort. And will the first sign of “weakness” lead to programs being abandoned? The product profile discipline inside of an R&D organization to know when to double-down and when to fold is paramount in such a hyper-competitive space.
Second, clinical trial enrollment and execution will be hugely competitive in light of all of the projects all attacking the same immunotherapeutic nodes. As noted, there are more than a 1000 clinical trials ongoing in the checkpoint space today; this probably represents 50-100K cancer patients in these trials now or soon (many are beginning recruitment). Cancer patient trial enrollment has always been challenging, but this bolus of activity will raise the issue to critical status. It’s fair to say there’s already a massive bottleneck around access to patients in the top cancer centers: it’s hard to imagine the most well-known academic medical centers in cancer, like MSK, Dana Farber, and MD Anderson, each running a dozen different PDx antibodies through clinical trials with their investigators. Furthermore, with approved checkpoint agents on the market, why would a physician recommend a trial where a patient might not get a PDx antibody (where the comparison might be to best standard of care chemo)? Already most trials today involve PDx plus or minus standard of care (SoC), rather than purely versus a conventional regimen; this is great for patients but makes differentiation all that much harder for the late entry products. Beyond enrollment, other aspects of clinical trial execution also pose new challenges: defining response rates in a world where RECIST tumor size and burden endpoints may no longer valid. Defining new immunological response rates, and how those predict overall survival, will be important (and are the subject of lots of current thinking). All of these aspects will conspire to make the clinical trial environment a hugely challenging operational task: big global footprints with lots of capital and clinical expertise, and broader leverage around privileged access clinical trial sites, will become even more important as a defining competitive advantage for cancer-focused drug companies.
Third, distinguishing signal from noise in the combination space will become increasingly difficult. If a PDx therapy alone delivers X% response rate in a specific cancer setting, and it can be increased in combination to by another 10-15%, that’s a huge win for patients. I was struck at ASCO by the incremental improvements (and material to patients) seen with a wide variety of combinations. The CHECKMATE 012 study’s combination of Yervoy and Opdivo demonstrated a nice bump in overall response rate versus monotherapy in NSCLC (i.e., confirmed ORRs moved from 23% to 47%). The addition of a MEK inhibitor to PDL1 therapy in colorectal cancer, a “rational combination”, demonstrated a very nice bump in activity. Trials ongoing with OX40 and CD137 reported early intriguing combination data (safer!), as well as with IDO/TDO approaches, STING, etc… But what happens if a large number of the 100+ different combination regimens today (e.g., PDx plus every other I/O target and chemo stack) deliver an incremental improvement in response rate in different settings? How do clinicians chose between them for their patients, and how do companies and their investors allocate capital? With the sheer volume of studies underway today, and the likelihood that many “rational combinations” will deliver some incremental improvement over PDx foundational therapy, this signal-to-noise becomes a huge challenge. Certainly highlights the importance of patient selection markers and such, but discerning how to combine for best effect will take years of work and significant investment.
Fourth, pricing of these therapies and combinations will become a flash point issue. With this many similar programs all entering the market – in settings where they will be used in large part in combination – stacked pricing models are unlikely to work. When it comes to combinations of these cancer drugs, 1+1 will not likely equal 2 in the eyes of payers. In addition, will payors convince Pharma to compete on price: “offer your PDx with massive rebates/discounts, and we’ll use it as foundation therapy in our pool of cancer patients”. In many crowded spaces, like TNFs or statins, this happened somewhat in the past. But given the much larger price tags here, and the combination of expensive novel products, this is likely to become a space where winners have deep access to the payor channel and figure out how to do bundled pricing models. Could this be the first time (or nearly so) that Pharma actually competes on price in a class of therapies? In addition, with survival curves extending longer – which is fantastic – more cancer patients are likely to stay on more therapies longer. Hopefully payors are modeling out these assumptions appropriately so they aren’t caught off guard like many were with HCV cures recently.
These and many other macro issues raise a bunch of specific strategic opportunities/challenges for different industry stakeholders in the biopharma arena:
- The Big Five: Those with big I/O checkpoint and cell therapy positions (BMS, Merck, Roche, AZ, and Novartis) are obviously focused on solidifying their “foundational” positions in the space, and in privileged spots in the treatment line-up for different indications (e.g., getting into 1L NSCLC). Launching as many smart clinical trials as possible, and tying up with as many combination partners as possible, will be key to the strategy. Smart BD groups at these five will aim to create as many proprietary partnerships and combination-deals as possible during this gold rush period. Once the dust settles, it’s unlikely that five PDx’s will be used interchangeably – and a massive clinical data advantage is one way to win.
- The Rest of Big BioPharma: If you don’t have a compelling clinical-stage checkpoint program to anchor your franchise, you’ve probably missed the boat – it will take a massive strategic bet around a creative partnership with one of the other players to get into the game in a meaningful way. Getting access to a foundational PDx agent in a smart way (that allows you to achieve a return on your combination offering) will be critical. Leveraging your existing cancer portfolio in an array of meaningful combinations is one strategy (like combining targeted kinase inhibitors such as MEKi with someone else’s PDx’s), but unless you’ve done something creative on the deal front, that still leaves you with the issue of pricing when the foundational PDx is not yours (the 1+1 doesn’t equal 2 issue above). These players may want to consider leapfrogging into next generation platforms (e.g., oncolytic viruses, functional bispecifics, next-gen CART/cell therapies) or targets (e.g., TAMs/MDSCs), and then using them as quids to access PDx more broadly in their portfolio. Lots of strategic issues for this group to wrestle down. To be provocative with a thought experiment, a player without a PDx today could partner with a group of payors to co-fund development of a “biosimilar”equivalent PD1/PDL1 mAb and price it “at cost” to those payors. Put their combination agents on top of this. A free PDx offering could be combined for value. Hard to see how this would work practically (would get to market a decade or more after Optivo/Keytruda), but payors would respond well to a low-cost PDx – an interesting concept. More creative thinking will be needed if these players-without-PDx are to compete.
- Emerging biotech companies. The conventional checkpoint space, and more generally T-cell I/O approaches, are increasingly expensive poker game to play. If you are a small aspiring biotech – private or public – you will need access to prodigious amounts of capital to fund new programs into development in I/O. Robust funding sources currently exist for the many strong companies (e.g., Forty Seven, Gritstone, Surface), but the reality is that powering up multiple parallel Phase 1b/2a studies in different indications (or large molecular basket trials) requires “bigger than venture” pocketbooks, and skilled teams large enough to run them – typically not the domain of most emerging small biotechs. Further, negotiating access to combination drugs like Opdivo or Keytruda will be important to keep trial costs reasonable. Because of this, in the long game, finding deep-pocketed Pharma partners with big oncology franchises will undoubtedly be the best outcome for many. Figuring out how to preserve product rights in key markets to enable “going long”, or how to extract an attractive enough M&A premium, will be important for these biotech players.
- Startups and Venture Capital. Creating the next new checkpoint-focused biotech is probably not a great idea in light of the issues above – congestion, signal-to-noise, competition, and commoditization are a challenging market mix for startups to jump into. New companies in this arena need to be truly different – new modalities, totally new targets, unorthodox approaches. But novelty itself will not be enough; translational approaches that distinguish themselves early in development need to be in place, and they need to figure out how to access foundational therapies for combination early (and hopefully demonstrate it preclinically, however poor those models are). Importantly, they also face the critical challenge of talent recruitment. An immune-oncology savvy and experienced CMO or CSO is a very highly sought-after profile; probably one of the toughest to fill in the industry today. Thinking about the parterning landscape at inception will also be a key strategic element: which Pharma partner(s) “needs” this asset the most to complement their existing portfolio (of the Big Five, for instance), or anchor a new leapfrog move into the space (the rest of Pharma). Given the investment in the space, and the nature of cycles, it’s also likely important to recognize that when the music stops, startups will really want to have a partner (or have something that partners really need). For instance, if everyone has a PDx or CTLA4 program already, what gives you confidence that yours will be of interest? Very hard to see an investment thesis around those targets get traction today from savvy investors. A healthy venture creation process begins in part with the endgame in mind: how do you build a great company around great science that people will want – either public investors or Pharma buyers (or both, preferably).
Fueled by the transformational clinical data around both survival and durability in an increasing range of cancers, the I/O explosion in cancer therapeutics is without a doubt a fantastic thing for patients.
But, like all hyper-competitive areas, there will be winners and losers. How cancer companies plan for and react to the macro strategic issues they face in this rapidly evolving field will determine their fate. Indeed, the next five years will certainly be fascinating to watch.