By Jonathan Montagu, CEO and Co-founder of HotSpot Therapeutics, as part of the From The Trenches feature of LifeSciVC
Today marks an important point in our journey to patients as HotSpot Therapeutics announces its $100MM Series C financing. This infusion of capital will enable HotSpot to transition into a clinical stage company while supporting further pipeline and platform growth. Our progress as a company tracks with the success of the entire field of allostery that has included Relay Therapeutics’ demonstration of clinical proof of concept; the acquisition of Vividion Therapeutics by Bayer; and significant capital raises made by Exo Therapeutics, Scorpion Therapeutics and Frontier Medicines.
Systematic approaches to identifying and drugging allosteric sites are now within reach which is exciting for patients as historically intractable disease proteins yield to therapeutic intervention. In this blog, I wanted to reflect on some learnings from the past couple of years as we have built HotSpot from the ground up while pointing to new opportunities that are now ahead of us.
Pockets that matter
As readers will know, the dominant focus of small molecule drug discovery has historically been the substrate or active site, but such pockets can be inhospitable to drug candidates and selectivity can be hard to achieve. As drug hunters, we have strived to go beyond the active site, but unfortunately very few pockets on proteins are actually relevant to function.
How then can we find the pockets on proteins that matter? Typically this involves random screening. Traditional machine learning approaches do not help much here since there is a paucity of data to train models outside the active site. In-cell fragment screening and protein motion simulation can find pockets but there is no guarantee that these pockets are critical to protein function.
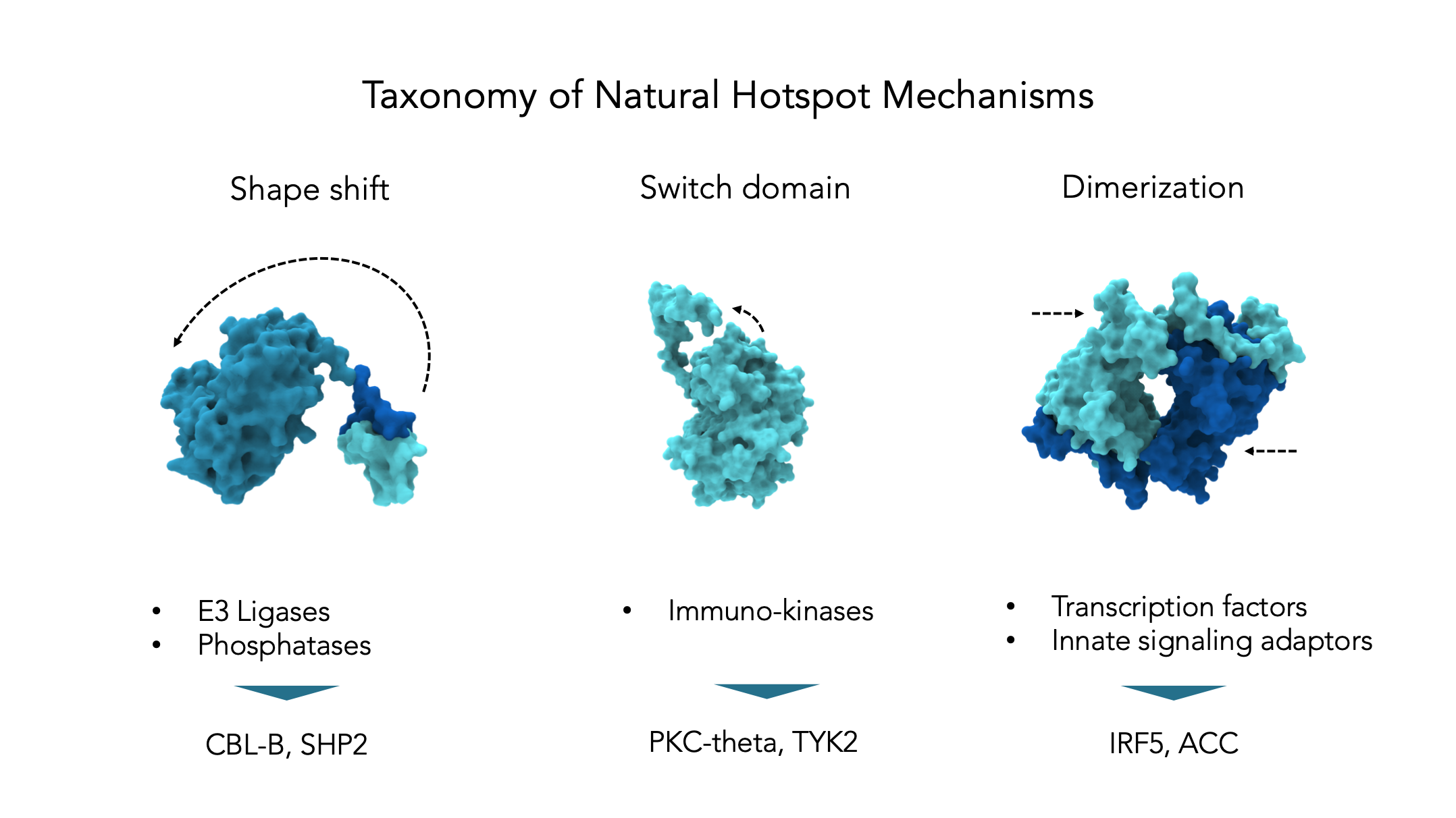
At HotSpot, we are solving this challenge through a sole focus on naturally-occuring pockets – called natural hotspots – that are decisive in the control of proteins in vivo. Natural hotspots encompass protein pockets and mechanisms used endogenously by the cell. This approach has allowed us to enrich for pockets, and in turn, small molecules that really matter to protein function. Moreover, since nature typically controls proteins with precision, natural hotspots tend to lead to highly selective modulators.
Using a suite of in-house developed artificial intelligence (AI) algorithms, we mine big and diverse datasets to build a picture of protein structure function that is solidly rooted in real-world evidence. Predictions are further amplified by looking across entire target classes. This has led to a new hotspot-focused taxonomy of proteins, where each category lends itself to a proprietary set of tools and chemistry that have been implemented inside HotSpot. Today, HotSpot has successfully progressed programs against all of these classes and is poised to select its first Development Candidates.
The Ghost in the Machine
Artificial Intelligence is a theme that has become ever more prominent in drug discovery but much of the excitement around small molecule AI relates to protein targets where there is a large body of existing evidence, for example, from previous big Pharma programs. Hotspot in contrast is uncovering new biological real estate and pairing this with novel chemotypes. Can AI really work at the frontiers of known biology and chemistry?
We are only just scratching the surface of what is possible but the answer is a clear ‘yes’. Our most successful applications of AI to date are:
- Relieving bottlenecks: through the algorithms described above we have identified about 1,500 targets with natural hotspot mechanisms. AI has been critical in helping us rapidly triage down to the cream of the crop targets that should enter our pipeline for wet lab prosecution. This ranges from natural hotspot prediction using algorithms that perform a type of facial recognition on 3D pockets, to feature detection amongst the screens of custom libraries directed at those pockets.
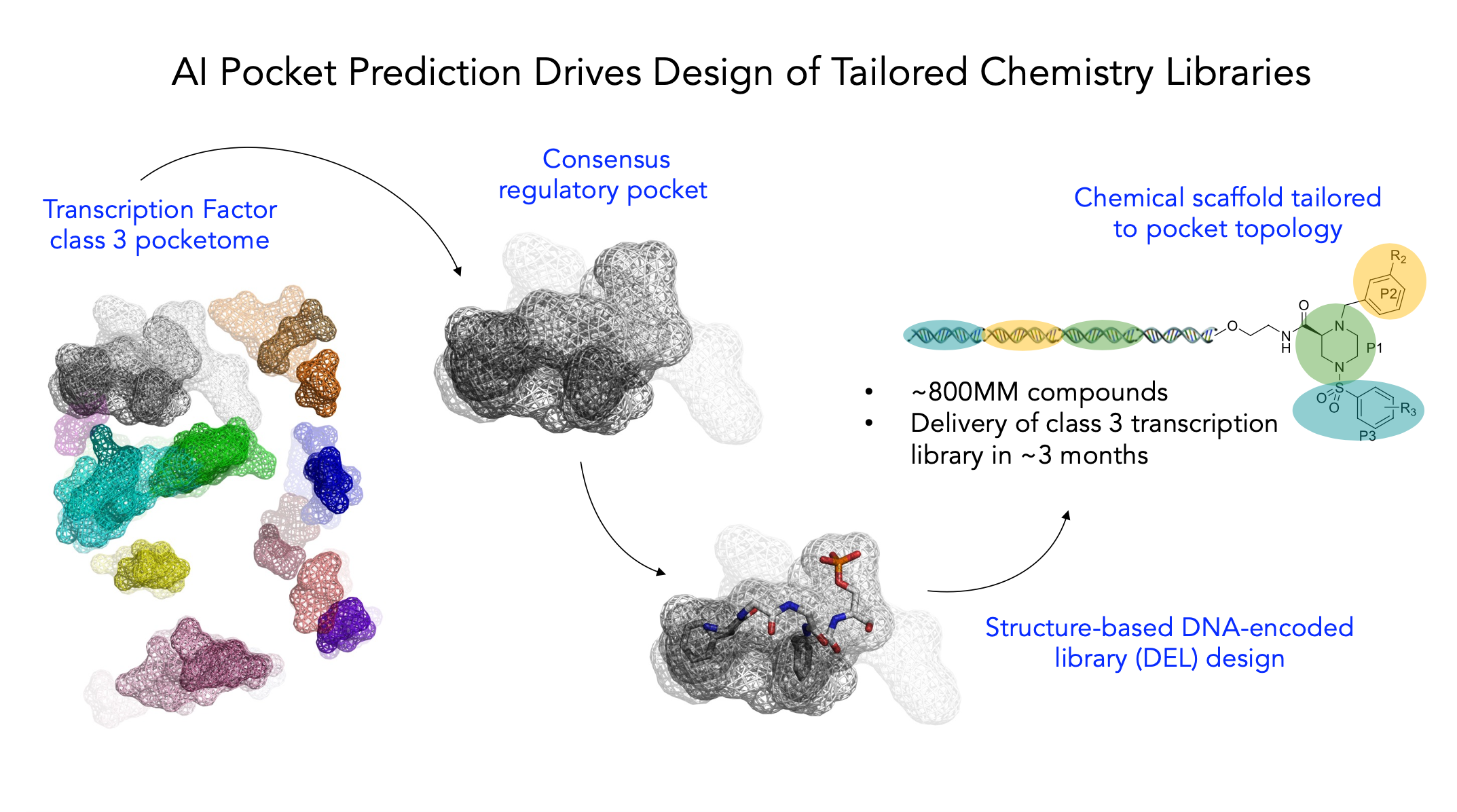
- Increasing program probability of success: machine learning allows us to make sense of the 60 million or so hits that we get from each chemical library screen. These datasets provide a huge volume of structure-activity relationship (SAR) data that would take traditional chemistry campaigns a decade to generate. Relay Therapeutics’ acquisition of ZebAI and our earlier acquisition of Macroceutics highlights the deep potential in this domain as the power of DNA-encoded libraries (DEL) meets structure-based drug discovery.
- Patient selection: As we now venture into patients, we have the ability to leverage rich patient data to link our understanding of the biology of our targets to specific patient subsets.
To illustrate AI in action, the figure below shows how we use custom machine learning algorithms to predict common regulatory pockets within a transcription factor class and then both design and screen a chemical library directed at those pockets.
Moreover, we have learnt that organization and culture are just as important as the underlying technology. In executing against AI within HotSpot, our team brings deep drug discovery experience to the problem – similar to our peer group companies – constantly with an eye to designing molecules that are going to go the distance. However, the cross pollination of the drug hunter mindset with the disruptive, software engineering perspective is what we strive to achieve at HotSpot. With this in mind, we have sourced exclusive rights to enabling technology from players such as Macroceutics, Molecular Discovery and idalab.
Sustaining competitive advantage
Our industry becomes ever more competitive and the success of one mechanism rapidly leads to follow-on competition. K-Ras, SHP2, EGFR, and BTK are good examples and the list goes on. New companies e.g. EQRx, and business models have arisen purely to exploit the fast-follower model while contracting with payers.
In order to maintain our leadership position in allostery, companies like HotSpot cannot be complacent since our competitors will erode competitive advantage. We need to be continually challenging ourselves to deliver both first- and best-in-class small molecules. Thankfully allostery provides all the raw ingredients to deliver molecules with exquisite potency, selectivity and drug-like properties. This is especially true for natural hotspots because they represent a privileged subset of allosteric pockets.
Sanjiv Patel, CEO of Relay Therapeutics, has been particularly thoughtful on the topic of competitive advantage which, to paraphrase him, is driven by:
- Riding the experience curve: delivering new allosteric medicines is technically very challenging and the industry leaders in allostery have successfully turned the crank multiple times. This leads to skill sets and scales of economy that can only be acquired over time, representing a large barrier to entry for new players.
- Exploiting non-commoditized technology: the technology that we use to identify and drug natural hotspots is unique to HotSpot and cannot be accessed through outsourced service providers. For example, we have access to the largest and most diverse chemical library designed around natural hotspots that delivers 10x hit rates of conventional chemistries.
- Leveraging a dominant position in a competitive labor market: as a company with a strong culture that is leading the way in natural allostery, we are able to disproportionately hire and retain the best talent in the most competitive labor ecosystems such as Boston and New Jersey. The creativity, drive and openness that our team brings to execution is a big differentiating factor.
To conclude, I am proud of what we have achieved over the past 36 months since it sets the stage for our first clinical trials and further growth of our pipeline and platform. However, I am also thrilled to be surrounded by such accomplished peer group companies that are also demonstrating the promise of allostery so convincingly. The next few years are going to be exciting as we deliver our allosteric medicines to patients.