By Deanna Petersen and Holly May, Chief Business Officer and Chief Commercial Officer, respectively, of AVROBIO, as part of the From The Trenches feature of LifeSciVC
Moving from traditional drug development and commercialization to gene therapy development and commercialization can be compared to moving from horses to cars as modes of transportation. That’s the analogy our CSO, Chris Mason, uses to describe the difference. Horses and cars were both game changers for transport in their respective eras (and cars continue to be), but both required different strategies to thrive.
Instead of building barns and growing hay and oats for horses, cars required more complex infrastructure like building gasoline stations, paving roads and training auto mechanics. Similarly, traditional drug development and gene therapy both work to bring us to our desired destinations – ultimately cures – but they require very different paths and planning to get there. The path to a successful gene therapy product is far more complex and nuanced than traditional drug development, and it requires people who can re-imagine every step along the way.
Commercializing gene therapy is still in the infancy stage, with only a handful of approved therapies “in-market” around the globe. Thus, similar to the infrastructure builds which were required as technology changed from horse drawn carriages to cars, we are still laying the groundwork to enable the shift from the traditional specialty pharmacy drug delivery model to a gene therapy archetype. This can not be a linear process, simply moving from clinical development to launch preparation. Due to the need to shift paradigms, gene therapy companies must lay the foundation for launch readiness years earlier than average. Those companies which figure out how to identify and solve “long-lead time items” win the race to market. Therefore, if you want to reach your ultimate destination delivering a potentially life-changing gene therapy to people living with a rare disease, you first need to know how to navigate the space.
The Nature of the Business
A gene therapy company operates using a unique business model due to the nature of the therapy. Gene therapies are designed to be a one-time therapy that can treat a genetic disease at its root cause – the altered gene. As a result, after being dosed, if the therapy works as intended, a patient is no longer part of the potential patient pool. Therefore, with every individual you (or your competitor) dose, your target patient population decreases by one…until you eventually get to very small patient numbers consistent with the incidence rate of the disease. Many diseases within the rare disease space have a high prevalent and low incident patient population; so as the target population decreases, the market potential decreases commensurately. Gene therapy companies generate value based on the number of patients they can treat, so there is a very narrow window within which to create, deliver and capture value.
The Top Line
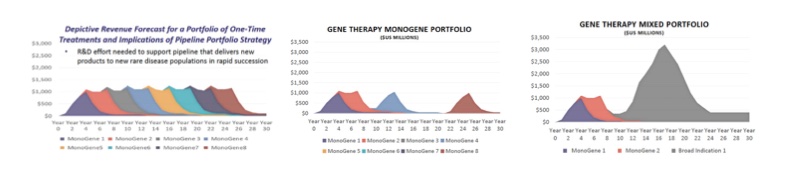
These factors all impact the projected revenue trajectory of a gene therapy business which focuses on rare disease prevalent populations – revenue and net income will ramp up in the first couple of years before slowly decreasing as the target market diminishes. As a result, to build a sustainable business and be financially successful, companies cannot stop at just one therapy – they need to continually develop and launch new therapies to keep top-line revenues somewhat stable. Our friends at Cello Health BioConsulting created a graphic a few years ago that helps illustrate what a successful gene therapy pipeline should look like.
Illustrative Gene Therapy Pipelines
Reprinted with permission from Cello Health BioConsulting
The first graph (left) has the next product launching as the prior product’s revenues are falling, keeping total revenues relatively consistent over time. This creates a predictive revenue forecast that supports a sustainable business. The middle graph shows what happens when there is a gap in your pipeline, leading to a period of time without a new product launch. Top-line revenues fall significantly – an event that investors do not like and one that does not set up for commercial success. Finally, the graph on the right shows what happens when you target a combination of small prevalent patient populations as well as a disease with a much broader indication. While ramps and declines are the norm in our business, it is easy to see why the importance of portfolio planning becomes amplified in the one-and-done gene therapy space. The lifecycle of a gene therapy can be dramatically shorter than that of a traditional chronic treatment, making pipeline planning all the more critical.
The Bottom Line
Understanding how gene therapy companies operate under this distinctive business model makes it easier to see why building a successful pipeline is ultimately a race against time. Speed is important in every biotech and pharma company, but even more so in a gene therapy company due to the “diminishing addressable patient population” nature of the business. What is good news for the patient – durability of affect with the promise of no need for future treatments – sets up a compelling business challenge to be first to market. The stakes become higher when operating in a competitive “one-and-done” development landscape. There must be a sense of urgency attached to every step of the clinical regulatory development plan for a gene therapy. Likewise, time plays an important role in planning for the commercial end-to-end patient support model; due to the unique nature of gene therapy, critical attention to detail needs to occur early to avoid potential downstream launch delays.
To be first, you need to operate with ‘smart speed’- a term we’ve coined at AVROBIO to describe working quickly and making calculated decisions to keep up your momentum. Every second counts in this space, and those who work with smart speed are always thinking about how to utilize each moment effectively. This is not only important for the business, but also for the patients and families waiting for new treatment options. Importantly, smart speed does not mean speed for the sake of speed or speed at any cost. Rushing forward without giving much thought as to what you are doing is more detrimental than beneficial. Smart speed requires making smart decisions based on good research and knowledge.
The gene therapy business model requires smart speed at every turn – and you have to understand that and embrace it in order to be successful and build a sustainable business. It is the model dictated to you by the nature of one-time therapies, and there is no debating it or swapping it out for a different business model.
Operating with smart speed is how you win.
The Model in Action
It pays to be on time in gene therapy, literally. If your company establishes an appropriate (and speedy) development timeline to market and stays firmly on that timeline, your company will create monetary value. The potential revenue stream from a product launch will be anticipated by investors, who will buy shares, which will drive share price up, thereby increasing the market cap of your company. And those future revenues can be discounted back to today’s value.
In the event your company is really good at implementing smart speed which facilitates quicker decision-making, which in turn shortens time to launch– say six months early – then, given the time-value of money, you will pull revenues forward and create value sooner. Increasing the net present value of a therapy is a well understood business concept, however, thinking about how this could affect a company’s overall valuation and potential share price is worth punctuating!
Being early to market has another advantage – if you are far enough ahead, your competition will likely drop off quickly. One of the problems with attractive markets is that they attract competition. You need to try to make it to market comfortably two or more years ahead of the competition to really reap the benefits of being early. No gene therapy company (or their investors) can afford to put time and money into expensive development of a gene therapy for a rare disease when they think a competitor will launch a few years ahead of them, especially if they don’t have a differentiated product. By the time they get to market, their target patient population would be significantly diminished.
There is a particularly big price to pay if a competitor gets to market ahead of you or comes to market within an estimated two-year window. If the former occurs, the competitor will start to deplete the prevalent population, leaving a far smaller addressable population to treat and putting you in a position where you must offer a significantly differentiated product compared to your competitor. If a competitor is close behind you, patients and doctors will naturally want to educate themselves on the pros and cons of each therapy, as they would with any new product coming to the market. However, with gene therapy, this choice is much more significant because of its one-time nature; there is no opportunity for a future “therapeutic switch.” Physicians and patients may prolong treatment decisions as they weigh the odds.
To Be Successful, Be First – How to Do It
Working with smart speed is the key to helping patients and families, dealing with the intricacies of the gene therapy industry, and operating successfully within this unique business model. If gene therapy development is like a car, the following smart speed principles are akin to best driving practices.
- Resource programs properly: Since speed and time are so critical in this business model, it stands to reason that companies should avoid having to prioritize or de-prioritize programs. Prioritizing programs means slowing something down in favor of boosting something else. Because value creation comes from being consistently on-time or early, de-prioritizing a program hinders its chance for success… and gives competition a chance to catch-up or surpass. In the real world, especially in a small company, sometimes it is necessary to disproportionately invest as the company raises money to support the necessary “cascading pipeline” illustrated above. When financing allows, the best strategy should always be to resource up! For example, if short on people, then hire more so you don’t burn out your workforce. Do everything you can to keep moving a program forward fast.
- Hire the right people: A company is only as good as its people. At AVROBIO, we look to hire candidates who love to drive programs forward by reimagining and reinventing how to go faster every step of the way. They are open to thinking differently and have experiences they can leverage in a variety of scenarios. They work shoulder-to-shoulder with their colleagues and continually ask, “What else can I (or we as a team) do to work faster?”
- Have a commercial mindset from the beginning: Adopting a commercial mindset at least four-to six years in advance of launch helps to ensure that a company delivers a therapy to market that meets the needs of the entire stakeholder ecosystem –Fulfilling unmet patient need, differentiating from the standard of care and demonstrating economic value to the health care system is essential for market success, and it requires a commitment from biotech companies to ask the right questions and make the right decisions early on, all with a commercial lens.
- Get ahead of long-lead time items: Investing in CMC, supply-chain and commercial capabilities early, enables preparation for the delivery complexities of gene therapy years ahead of approval. Companies need to conduct a detailed analysis of end-to-end administration at commercial scale, to identify barriers not apparent at clinical stages. Custom solutions, the creation of centers of excellence, optimizing the patient experience and follow-up all need to be clearly thought through. Additionally, educating physicians, patients, care-givers and payers/HTAs on the value proposition of gene therapy versus traditional standard of care is a multi-year critical investment due to the “newness” of the treatment modality. All these need to be done in parallel to product development so as not to prolong time to market.
- Think ahead and take smart risks: To build a sustainable gene therapy business, it’s important to always think about what comes next in your development pipeline. Keep enough potential therapies going in your pipeline so that when revenues start to decline from one, there’s another one launching to replace those revenues. Work to leverage the technology you’ve developed for one program for the next. Be judicious about the diseases you choose and don’t be afraid to take smart, well informed risks as a company.
Understanding your business model and what drives it, is critical for success. For gene therapy, the fundamental factor underpinning success is speed…also known as ‘smart speed’ at AVROBIO. It’s easy to see the value of smart speed at the individual level – it is beneficial to your team and helps you accomplish your tasks. On the company level, it can be harder to see, this is where leadership’s communication and reaffirmation of the corporate strategy comes in to play. You have one shot to be first to meet the needs of the patients you aim to serve– therefore every decision made, every second of every minute of every day, needs to be made to support that accelerated timeline.
#everysecond