By Abbas Kazimi, CEO of Nimbus Therapeutics, as part of the From The Trenches feature of LifeSciVC
In early 2009, when global markets were bottoming out, you can imagine what it felt like walking the halls of the JPM Healthcare Conference. Biotech felt fragile. Capital was scarce, pipelines were thin, and more than a few investors were quietly asking whether the venture model in drug discovery even worked anymore.
That was also the moment when a small group of firms, Atlas Venture included, were sketching a different kind of biotech on whiteboards with lawyers and scientists. Not a fully integrated pharma. Not a lab-heavy roll up. But a focused, small-molecule company built around chemistry, leading computational tools (Schrödinger in our case), and a virtual operating model that could survive volatility and still produce real drugs.
Nimbus was born out of that era.
We did not set out to build the biggest labs. In fact, Nimbus has never had its own labs. We set out to build the fastest learning engine.
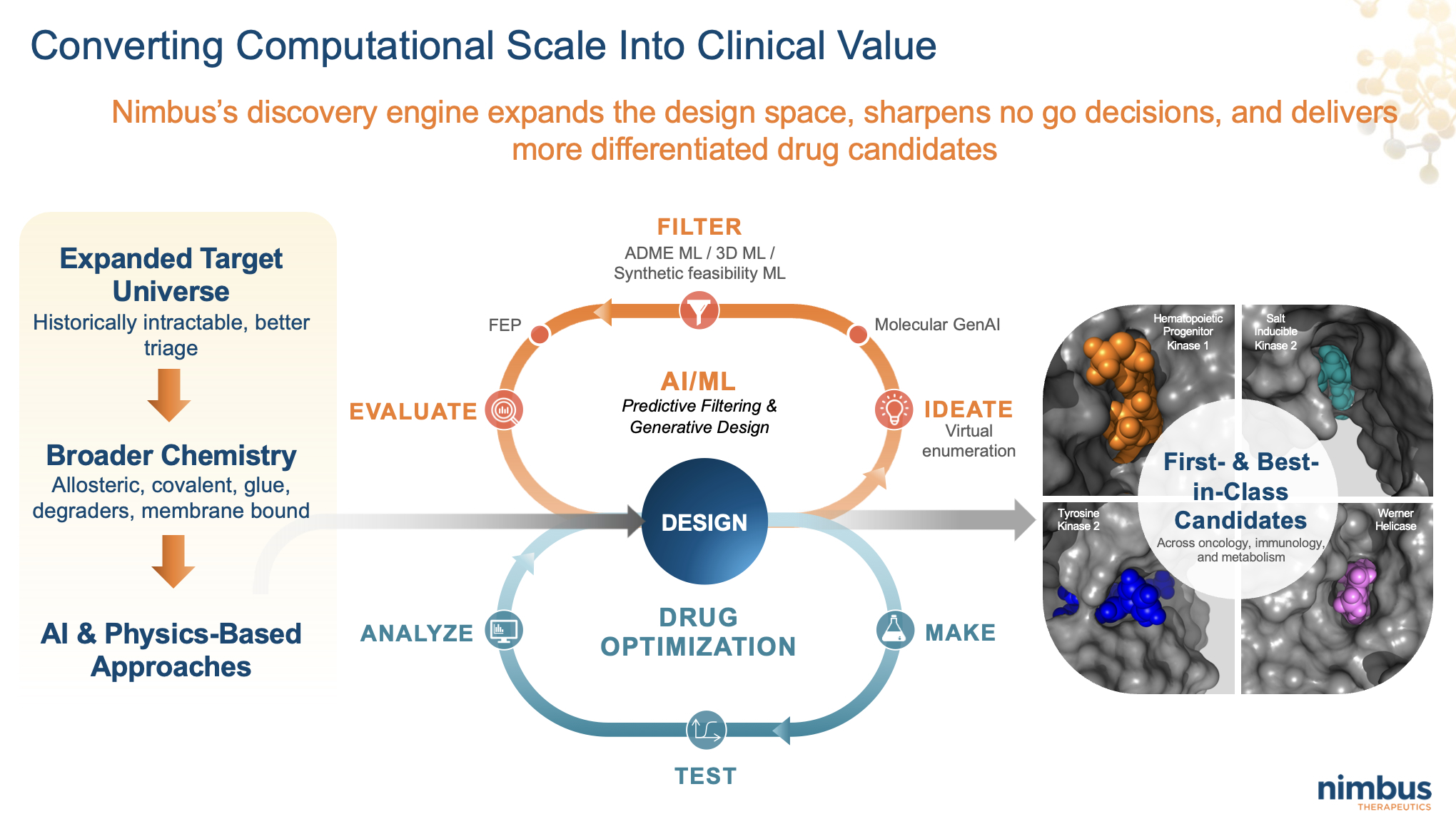
Sixteen years later, Nimbus has sold drugs to Gilead (here) and Takeda (here), partnered programs with Genentech (Roche) (here), Celgene (BMS) (here), and most recently with Lilly (again) (here, here), and built what we believe is one of the most consistent small-molecule engines in biotech. We did that by owning the Design–Make–Test–Analyze loop, using global partners to execute at scale without ever giving up control of what mattered.
Today, DMTA at Nimbus is concrete and central to the business. We design molecules on computers. We make them through CRO partners. We test them in proprietary, bespoke biological screening cascades built for each target. We analyze the data and feed it directly back into the next design cycle using integrated AI and physics-based approaches.
That lived experience is why a recent essay on geopolitical risk in biotech sparked conversations about whether Western pharma may have inadvertently funded a competing biotech ecosystem by treating preclinical biology as a commodity.
The piece argues that outsourcing execution inevitably leads to outsourcing learning, leaving Western biotech’s licensing assets from foreign ecosystems that their own capital, years before, helped train.
Our experience suggests another path is possible. Nimbus continues to make compounds that are fully ours: the molecules, the data, and the learning.
The IP sits in the molecule, assigned cleanly to a designated subsidiary. More importantly, the learning loop lives inside the company, giving us repeatability and conviction.
It is also worth acknowledging how much the landscape has changed. Chinese biotechs and TechBio companies have made real progress, particularly in medicinal chemistry efficiency and the rapid advancement of me-too clinical candidates. Timelines for these “follow-on” programs have compressed, execution has improved, and competition has increased.
But that progress has been uneven. Generic medicinal chemistry and early clinical execution are increasingly commoditized. What remains difficult, and far less transferable, is preclinical biology and the novel chemical equity to crack it open: identifying high-value targets, building disease conviction, generating unique chemical tools, and designing experiments that separate signal from noise before patients and capital are put at risk. This is where many of these models remain weakest today.
Nimbus has always invested there. As execution becomes more intensely competitive, biological insight becomes the differentiator.
DMTA Is Not New. Compression Is.
The best small-molecule teams have always worked in loops. Form a hypothesis. Make a molecule. Test it. Analyze the data. Decide what to do next. That loop has always existed inside strong core teams, from Merck in its small molecule heyday of the 1980-1990s to today’s innovative “smart chemistry” firms like Nimbus and others.
What AI and physics-based methods have changed is not the existence of the loop, but its speed and resolution. They let you explore more chemical space, filter more aggressively, and arrive at better experiments faster. Nimbus has been integrating generative design, free-energy perturbation (FEP), Absorption, Distribution, Metabolism, and Excretion (ADME) and pharmacokinetics (PK), prediction, and structure-based modeling into daily decision making for years.
While we never had a ‘platform’ the loop was always the engine – our engine. AI now compresses it.
Asset-Light Does Not Mean Learning-Light
There is an important distinction between outsourcing execution and outsourcing learning. The concern that companies might be training their competitors is valid when the learning loop itself moves offshore.
Shared infrastructure doesn’t inherently weaken drug design capabilities. This is exactly what cloud computing did for software. No startup built its own data center. Yet the winners still owned their code, their architecture, and their product decisions. Shared infrastructure did not weaken those companies. It strengthened them by allowing talent and judgment to concentrate where they mattered most.
Nimbus fits cleanly into that logic.
Not every startup needs to build a full-stack lab. What matters is maintaining control of the learning loop. CROs and CDMOs are execution layers. Nimbus owns the hypotheses, the molecular designs, the prioritization logic, the data integration, and the go or no-go decisions.
The learning stays inside the company. That distinction matters.
Rational Capital Structures, Intentional Strategy
Asset-light models emerged partly from venture financing and governance realities. That is true. But for Nimbus, the virtual model was not simply a response to capital constraint. It was an intentional strategy that allowed us to survive multiple capital cycles, preserve optionality, compound learning over long horizons, and partner repeatedly from a position of strength.
Because we were not anchored to fixed infrastructure or a single domain of expertise, we could adapt organically to the most compelling science, whether in immunology, oncology, cardiometabolic disease, or liver disease. The absence of in-house labs was not a limitation. It created freedom to pursue programs opportunistically and strategically, guided by data rather than structure.
Sixteen years of outcomes suggest this can be a successful approach when executed with discipline.
Sovereignty Lives in Decisions
There is a real and important question about sovereignty and resilience at the industry level. But startups are not meant to be sovereign infrastructures. Building and maintaining domestic capacity at scale for long-term industrial resilience is a national welfare problem, typically carried by governments, strategics, or shared platforms.
Nimbus’s model fits that reality. We do not aim to be a sovereign infrastructure. We aim to be a sovereign decision engine.
That is exactly the role startups should play. Focus on judgment, learning, and conviction, while leveraging shared execution capacity to move faster and stay flexible.
Coming Full Circle
Biology is not semiconductors. It is messy, much more complex, and deeply human. Value accrues in layers. Execution and data generation sit at the base. Hypothesis generation, molecular design, and the integration of weak signals into coherent decisions sit at the top.
Nimbus has always invested at the top of that stack.
As I reflect on the JPM Healthcare Conference now in 2026, nearly sixteen years after those early conversations, what struck me most was how familiar the dialogue still felt. At meetings, dinners, and side conversations, the focus was not on infrastructure or geopolitics.
It was on the quality of the drugs. The molecules. Whether they would matter for patients.
That is where our IP lives. That is where our sovereignty lives. Nimbus was built for that world long before it was fashionable to describe it that way.
Many thanks for the contributions and editorial advice from Cindy Fung (Nimbus head of corporate affairs), Peter Tummino (President, Research & Development), and Bruce Booth, (Partner, Atlas Venture).