By Arthur Tzianabos, CEO of Lifordi Immunotherapeutics, as part of the From The Trenches feature of LifeSciVC
As biotech companies tackle the escalating time and cost of clinical trials, the decision to conduct early-stage, proof-of-concept studies outside the U.S. is becoming more common. While this approach is often faster and sometimes cheaper, it should not be the sole driver. Instead, there should be a thoughtful, deliberate set of strategic discussions within the company and its board that considers many factors and requires a careful benefit/risk assessment.
Here, I want to highlight some of the considerations and challenges (expected and unexpected) for those of you who are contemplating this endeavor. As Bruce Booth noted in his 20 year reflections last fall, “the single most important value creation activity in biotech is excellent clinical trial execution ”─so getting this right is critical.
Conducting clinical trials abroad will impact every function in the organization. Some things can be anticipated, such as managing different time zones, language barriers, diverse patient populations, materials and drug supply, and supply chains. However, it is the unexpected issues that can have the greatest impact on the success of the trial and in some cases the entire company. This harkens me back to the old mantra, “Don’t let the trial fail the drug,” a truth that may be even more fitting when conducting clinical trials in other regions of the world.
Here, I have collected insights from my own experiences and from a few of my biotech colleagues to provide additional perspectives. Deciding which examples to include was not easy. Capturing the ones that can be anticipated but may be underestimated, as well as the unexpected and costly ones, should serve as good reminders and a heads-up for all of us.
Speed and Cost of Trials
The cost of ex-US clinical trials is generally considered to be much less, somewhere on the order of 30-50% cheaper (though not 70-80% as some might think). I have yet to meet a company that conducted trials outside the U.S. and correctly budgeted for travel and other ancillary costs. The frequency with which employees will need to visit clinical sites, key vendors or manufacturing plants can be surprising. Unforeseen situations will arise and the unanticipated, last-minute flights and extended stays are expensive. Additionally, a company may need to provide standard of care (SOC) medicines that are often not available in other countries, and/or offer post-treatment therapies, both of which drive up clinical trial costs. Establishing an in-country presence, whether it is someone ‘parked’ there or hired as your own company representative, may seem like a luxury. However, it is a highly recommended strategy that ultimately can save time and money. When running trials in the EU, companies also need to sponsor a qualified person to ensure the quality of materials and facilities meet GMP standards. Local regulators will examine materials and want either compendial materials or locally sourced ones. A company will need to know and prepare for this ahead of submitting the required investigational product paperwork. Since most clinical plans evolve later on, it is critical to cast a wide net in the process.
Tax incentives for conducting clinical trials in certain territories can be very attractive. Understanding the details of these is important because they can impact a company’s cash runway and change fundraising plans. Several companies have recounted experiencing ‘tax breaks by two offices.’ This relates to a particular region of the world where one business office distributed the money from tax incentives while another ensured all requirements were met. It became clear that the offices operated independently and didn’t always communicate the same information. As a result, the money received was far less than anticipated, or a significant portion of the money needed to be returned.
These are just a few of the cost considerations when deciding to conduct clinical trials outside the U.S., however, there are other important factors that might influence this decision.
Patient Populations
Studying a more geographically diverse patient population in a clinical trial offers broader safety and efficacy evaluation as well as an opportunity to establish relationships with a global network of regulators and key opinion leaders. This also helps pave the way for broader commercialization beyond the U.S. Accessing patients might be easier, faster, and less expensive in some places. Accounting for ethnic, genetic, cultural, lifestyle differences, pre-existing conditions and environmental exposures is also required. The “all medicine is local” aspect to this can become apparent during screening and enrollment. In one particular example, it wasn’t until a company had designed the protocol, selected a clinical research organization (CRO), and identified clinical trial sites, that it learned about a genetic mutation native to the area at reasonable background levels that it could predispose healthy participants to screen-fail at a much higher rate. Fortunately, the trial hadn’t started, but it required a protocol amendment, ensuring that a local lab could perform a non-standard test, and increasing the number of patients to be screened.
Holding frequent and detailed meetings with CRO leadership and the teams assigned to your study, clinical investigators and trial site coordinators before a protocol is finalized is critical to avoid unexpected issues like this. It could have saved another company from learning too late that the assay it developed to measure an enzyme involved in fat metabolism did not perform well in a local population whose diet preference was eating sausages at every meal. Companies need to understand how local populations live, receive care, and how those factors might influence trial outcomes.
The Practice of Medicine and Clinical Endpoints
As a condition of approval, FDA typically requires that companies demonstrate that the clinical data generated ex-US is representative of the patient population and the practice of medicine in the U.S. While healthcare is managed and reimbursed differently throughout the world, the details are often underappreciated. Depending on the region, health records might be unavailable, incomplete, or unreliable, particularly where care is not tightly monitored. Assessing health and compliance with SOC can be challenging.
Relying solely on CROs to ensure protocols are being followed and all aspects of a trial are well managed is one of the biggest mistakes companies can make. While CROs and trial sites are responsible for overseeing compliance, one company running an ex-US trial in a region where compliance is particularly challenging decided to administer a test to determine whether study participants were taking the SOC medicine during the trial screening period. This would help determine patients who are likely to be compliant during the study and minimize the risk of enrolling the wrong patients. Companies might also pay for crossover treatments, especially for Phase 1 trials, thus providing SOC treatment for a full year post study.
To complement CRO efforts, some companies choose to have ‘boots on the ground’ for key trial events such as first dose(s), especially for first-in-human (FIH) studies. It is expensive but often yields the greatest ROI. As issues arise, extended travel can’t be avoided. It drains employees and organizations when people are away for weeks or a month at a time, particularly for emerging biotechs who are stretched thin and people wear multiple hats. The best advice is to identify potential needs ahead of time. This alleviates pressure, maintains strong relationships inside the company and with ex-US partners, and minimizes clinical trial risks longer term.
Building trust with clinical trial partners in certain foreign countries can be harder and take longer. It is also not something that should be left solely to the CRO. Getting to know the people, cultural norms and communication preferences helps to bridge the distance. It can take months to develop strong enough relationships in some countries compared with others where Zoom meetings are enough to garner the trust needed to get clinical trials up and running.
Establishing clinically meaningful endpoints may vary across regions. There can be different measurement ranges that reflect clinical practice and these complicate data analyses. Understanding exactly how endpoints will be measured and patient outcomes will be recorded is essential for trial success. Language barriers, treatment regimens, diagnostics capabilities ─including imaging and videos, and lifestyle differences can all lead to inconsistent interpretations and assessments. One biotech company with global trials involving over 7,500 patients and ~600 clinical trial sites had to align on big-ticket items with multiple regulatory agencies, clinical sites, and ethics committees across regions. Country-specific amendments were expected and accepted.
Recently, FDA has asked at least two companies for a placebo arm requiring some form of an invasive surgical procedure for their pivotal trials. In each case, these are in rare disease patient populations that require delivery of drugs to the central nervous system. In one case (with which I am familiar), several European countries deemed this approach unethical, which required additional placebo patients to be added to the study to perform a less invasive surgery. The lack of alignment between regulatory agencies continues to be a major challenge.
Legal and Regulatory Considerations
As noted above, regulatory requirements vary by country and region, but a clinical development strategy should be global from the outset. Aligning study design and protocols with appropriate regulatory expectations and operational realities is critical to avoid delays, cost overruns, and complicated data interpretations. Regulatory filings outside the U.S. are often less extensive than full investigational new drug (IND) submissions and review timelines may be shorter, however early engagement with the FDA is important. Even when initial clinical proof-of-concept data will be generated ex-US, conducting a pre-IND meeting before completing GLP/GMP toxicology studies helps ensure the nonclinical package aligns with their feedback and meets FDA expectations. Unfortunately, some companies have encountered significant delays presenting positive ex-US clinical data to the FDA and learned afterwards that additional toxicology work would be required.
International trials must also comply with country-specific legal standards governing data privacy, confidentiality, and cross-border data transfer. In the EU, data protection regulations differ significantly from HIPAA requirements in the U.S. and require companies to appoint a Data Protection Officer to oversee compliance. Understanding the requirements and incorporating them into clinical trial applications and site selection decisions can prevent delays, penalties, and incurring unexpected costs later on.
Manufacturing and Supply Chain Logistics
It is considered good practice for Chemistry, Manufacturing and Controls (CMC) company personnel to be present for drug production runs. While this requires additional time and expense, experienced operators believe this is non-negotiable. At my company, Lifordi Immunotherapeutics, I had a senior member of my CMC team observe our first production runs in Scotland. Due to a number of issues, my colleague spent several late nights eating dinner from an all-night gas station food market but was thankfully able to manage through the process before it became a major problem for us.
Supply chains and shipping logistics for ex-US trials are complex. It can take a village ─or large network of reliable vendors ─to achieve success. In one instance, a clinical site near a war-torn territory necessitated that materials be flown to a neighboring country and driven across the border to the clinical trial site. Patient samples needed to take a similar route for proper analyses. Maintaining temperatures for drug product, sample storage, and delivery was a top priority. Companies routinely manage these logistics by keeping ahead of things such as new labelling requirements ─a situation that is known to cause delays and incur further expense. Today, rapidly changing import/export rules and imposed tariffs are further complicating supply logistics. New regulations often impact regulatory filings and while the magnitude of changes can be difficult to predict, the potential time and cost to comply must be budget.
Destinations for Ex-US Clinical Trials
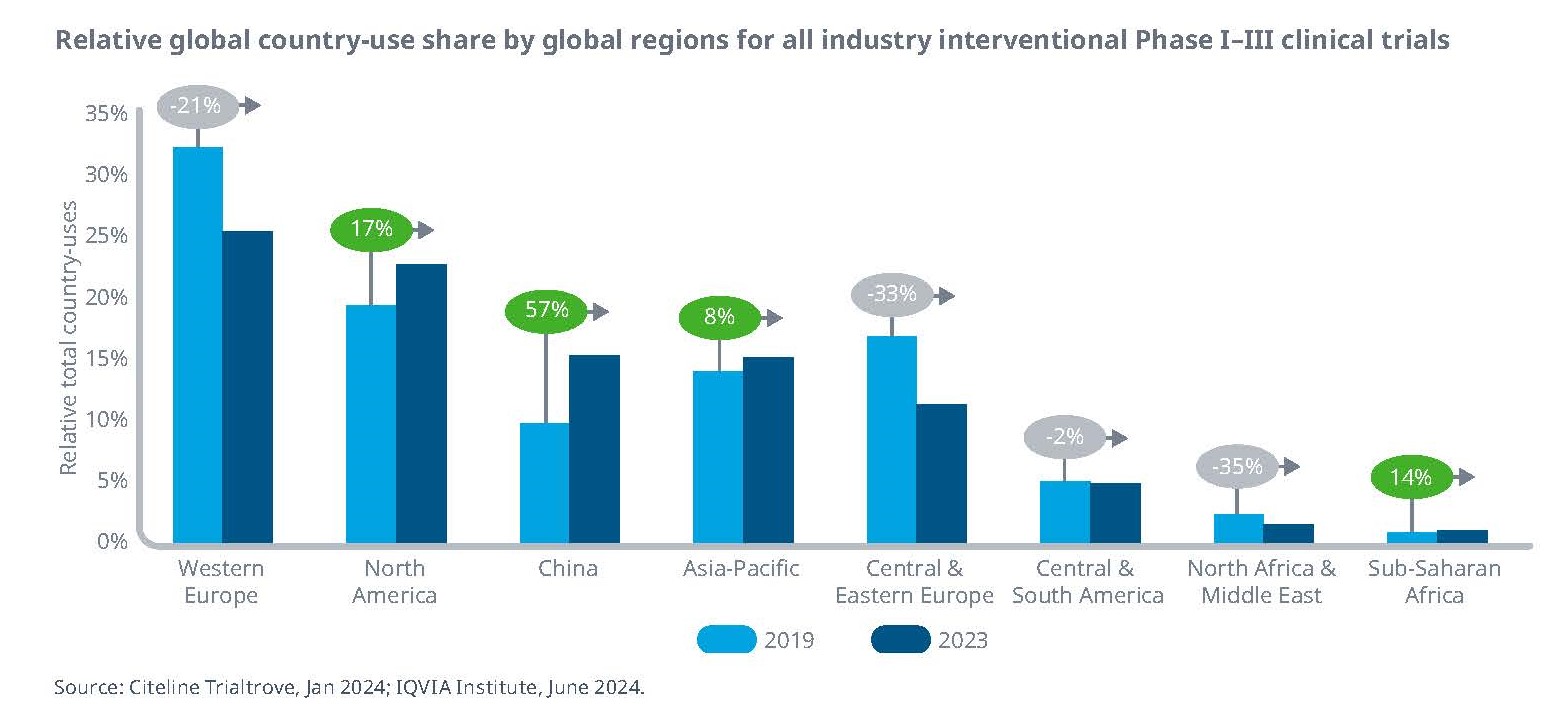
Governments, pharma companies, and other industry partners continue to invest in building critical infrastructure overseas including training and creating substantial talent pools. For many years, conducting clinical trials outside the U.S. included locations in Europe, Australia, Japan, and Latin America. Today, Asia-Pacific is the fastest growing region with major investments in China made by pharma companies including a $15B investment by AstraZeneca and a ~$1B investment by Pfizer ─both through 2030, and more recently, a $484M investment in South Korea over the next five years by Roche.
China has increased its relative share of clinical trials by 57% from 2019 to 2023, and in 2024 it was the third most utilized region mainly due to its large patient population demanding new therapies, technological capabilities, cost-effective solutions, and favorable regulatory policies.
While this increase was mostly driven by the explosion of clinical work from China-based companies, trial recruitment in China can also be 5-10 times faster than it is in the U.S. and averages one patient per month for some indications. For these reasons, China is increasingly a topic of discussion by Western companies for FIH studies.
Currently, most US companies are doing FIH trials in Australia (including Lifordi), New Zealand, Belgium or the Netherlands. Timelines remain rapid in these countries. For healthy participant studies, it takes approximately 2-3 months from final protocol and investigator’s brochure through to dosing first patient. SAD/MAD studies can be completed with 4-week follow-up within about 6 months. Healthy participant data is also readily accepted by the FDA.
Conclusion
As companies weigh the risks and benefits of conducting clinical trials outside the U.S., rapid data generation and potential cost-savings often rise to the top. Other substantial advantages include access to diverse populations and broader access to innovative medicines for patients. Yet these benefits come with expected and unexpected challenges. Companies that invest time and resources to develop local knowledge and build strong relationships are far better positioned to achieve success and avoid costly setbacks. As the old mantra reminds us, careful planning and execution is key—because no company wants the trial to fail the drug.